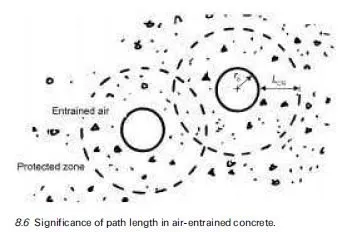
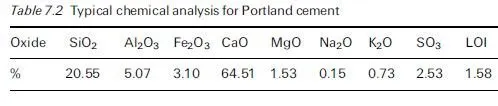
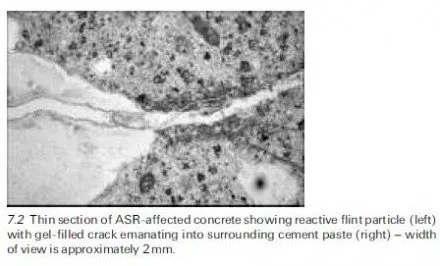
Thaumasite is normally formed at temperatures below ca. 15ëC and preferably at 0±5ëC, which seems to be the optimal temperature range because the solubility of silicate, though very small (in ppm levels), is relatively greater within this tem- perature range. A through-solution mechanism is more likely to arise under such conditions and its existence would explain the general slowness of the thaumasite- forming reaction, especially in the first few weeks and months. However, there is circumstantial evidence that, in some instances, thaumasite might actually be produced when the temperatures are above ca. 15ëC (Bensted, 2001).
The possibility of thaumasite appearing at more elevated temperatures had not been definitively proven (Luke, 1998; Bensted, 2001), but could (theoretically at least) arise at higher temperatures under pressure, when an arrangement of [Si(OH)6]2ÿ groups might be sufficiently stable to promote formation of thaumasite. Such a situation would be most likely to be facilitated if crystal- lisation pressures were sufficiently high to enhance the stability of a transition state intermediate of six OH- ions surrounding the highly polarising Si4+ cations for sufficient time to allow thaumasite to emerge as a deterioration product. Surveys of thaumasite sulfate attack (Diamond, 2003; Sims and Huntley, 2004) suggested that thaumasite sulfate attack might occur at temperatures above 15ëC but there was no definitive evidence given by them. The first conclusive evidence for thaumasite forming above 15ëC revealed that small quantities of thaumasite had been produced after 720 days at 20ëC by the direct route. This was contrasted with the analagous situation at 5ëC when extensive formation of thaumasite had been observed (Malolepsy and Mro z, 2006).This was in accord with an earlier prediction (Bensted, 2001).